
C57BL/6-Cd40tm1(CD40)Bcgen/Bcgen • 110009

| Product name | B-hCD40 mice |
|---|---|
| Catalog number | 110009 |
| Strain name | C57BL/6-Cd40tm1(CD40)Bcgen/Bcgen |
| Strain background | C57BL/6 |
| NCBI gene ID | (Human) |
| Aliases | p50; Bp50; CDW40; TNFRSF5 |
CD40 is a member of the tumor necrosis factor receptor (TNFR) superfamily and is primarily expressed on antigen-presenting cells, including dendritic cells, B cells, macrophages, and monocytes. Interaction between CD40 and its ligand CD40L (CD154) plays a central role in adaptive immunity by promoting antigen presentation, cytokine production, and T-cell priming. Due to its immunostimulatory function, CD40 is a key target for cancer immunotherapy, vaccine adjuvant strategies, and immune modulation research.
In CD40 humanized mice (B-hCD40), the endogenous murine Cd40 gene is replaced with the human CD40 coding sequence to enable species-specific expression of human CD40 under native regulatory control. This humanization design supports translationally relevant evalsuation of anti-human CD40 biologics by allowing therapeutic engagement with human CD40 in an in vivo immunocompetent setting while eliminating interference from murine CD40.
Validation data confirm the successful establishment of CD40 humanized mice (B-hCD40), supporting their use as a preclinical model for pharmacodynamic, efficacy, and safety assessment of CD40-targeting therapeutics.
Key Advantages
Validation
Application: CD40 humanized mice (B-hCD40) are used for in vivo evalsuation of CD40-targeted antibodies, immune agonists, and combination immunotherapies by enabling human CD40-specific immune activation, antitumor efficacy assessment, and translational safety profiling.
The exons 2-7 of mouse Cd40 gene that encode the extracellular domain were replaced by human CD40 exons 2-7 in CD40 humanized mice (B-hCD40).
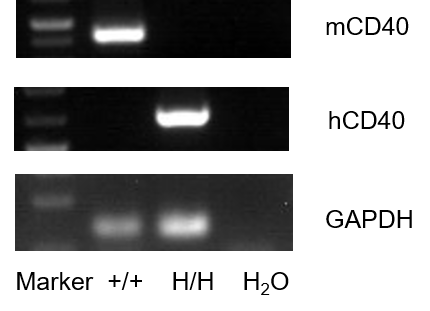
Strain specific analysis of CD40 gene expression in WT and B-hCD40 mice by RT-PCR. Mouse Cd40 mRNA was detectable in splenocytes of wild-type (+/+). Human CD40 mRNA was detectable only in H/H but not in +/+ mice.
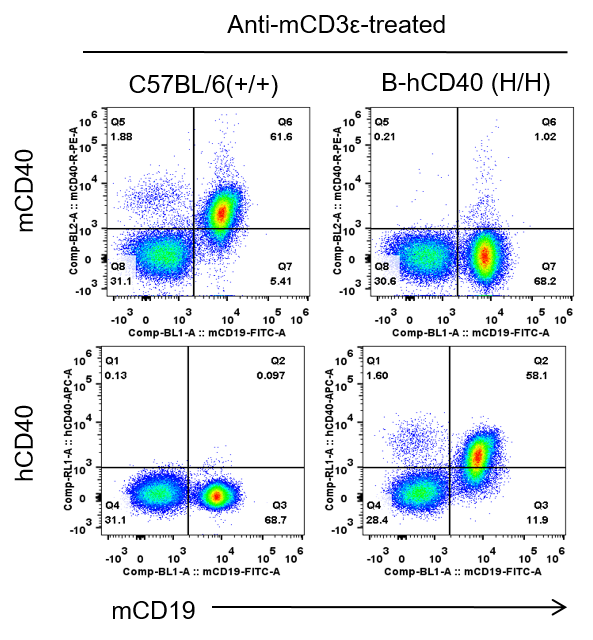
Strain specific CD40 expression analysis in homozygous B-hCD40 mice by flow cytometry. Splenocytes were collected from WT and homozygous B-hCD40 (H/H) mice stimulated with anti-CD3ε in vivo (7.5μg/mice), and analyzed by flow cytometry with species-specific anti-CD40 antibody. Mouse CD40 was exclusively detectable in WT mice. Human CD40 were exclusively detectable in homozygous B-hCD40 but not WT mice.
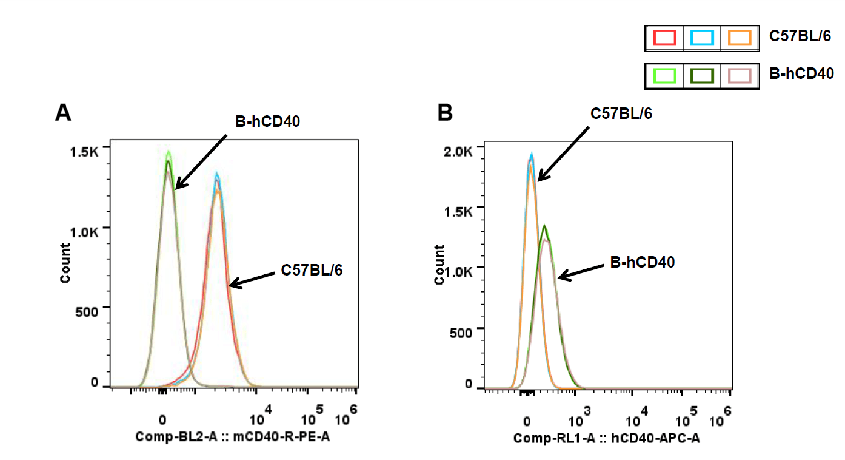
Strain-specific CD40 protein expression in splenic B cells of CD40 humanized mice (B-hCD40) was evalsuated by flow cytometry. Splenocytes were isolated from wild-type C57BL/6 mice and homozygous CD40 humanized mice (B-hCD40, H/H) and stained with species-specific CD40 antibodies. Flow cytometric analysis demonstrated that mouse CD40 was readily detectable on B cells from wild-type C57BL/6 mice but was absent in CD40 humanized mice (B-hCD40) (A). In contrast, human CD40 expression was exclusively detected on B cells from homozygous CD40 humanized mice (B-hCD40) and was not observed in wild-type controls (B). These results confirm the successful and specific replacement of endogenous mouse CD40 with human CD40 in the splenic B-cell compartment of CD40 humanized mice (B-hCD40).
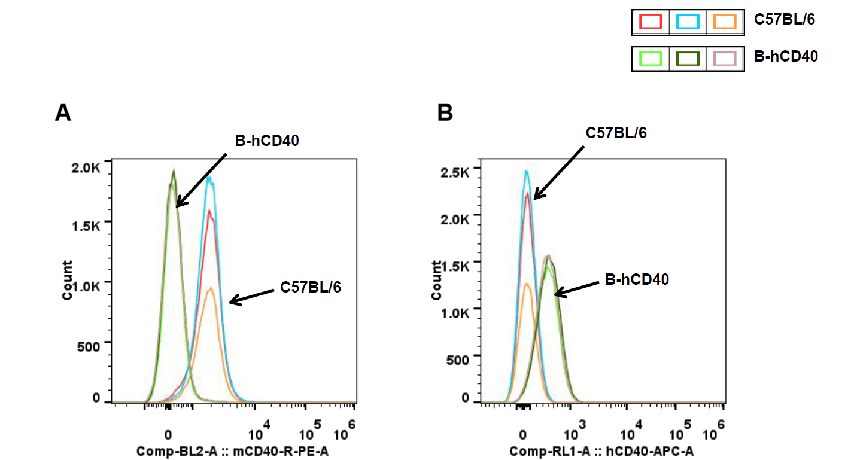
Strain-specific CD40 protein expression in peripheral blood B cells of CD40 humanized mice (B-hCD40) was analyzed by flow cytometry. Peripheral blood samples were collected from wild-type C57BL/6 mice and homozygous CD40 humanized mice (B-hCD40, H/H). B cells were gated and analyzed using species-specific CD40 antibodies. Flow cytometry results show mouse CD40 expression was detectable only in peripheral blood B cells of wild-type C57BL/6 mice) (A), whereas human CD40 expression was exclusively detected in peripheral blood B cells of homozygous CD40 humanized mice (B-hCD40) and demonstrated that was absent in wild-type controls) (B).
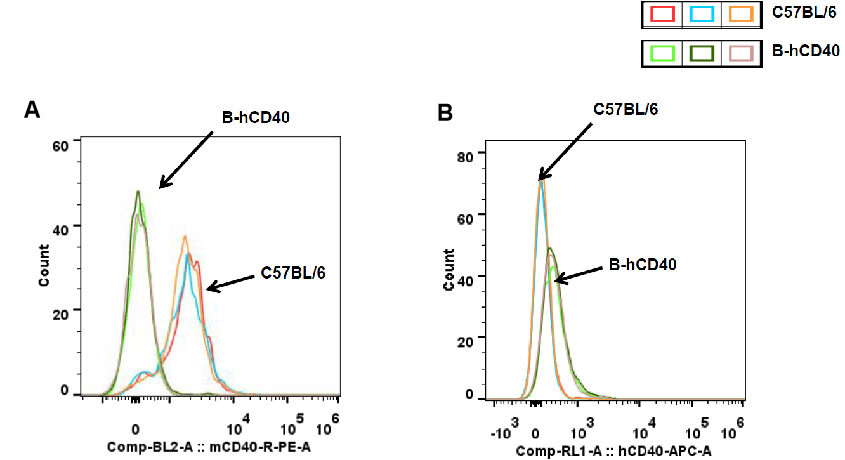
Strain-specific CD40 protein expression in splenic dendritic (DC) cells of CD40 humanized mice (B-hCD40) was analyzed by flow cytometry. Splenocytes were collected from wild-type C57BL/6 mice and homozygous CD40 humanized mice (B-hCD40, H/H), and DC cells were gated and analyzed using species-specific anti-CD40 antibodies. Flow cytometry results demonstrated that mouse CD40 expression was detectable in splenic DC cells of wild-type C57BL/6 mice) (A), whereas human CD40 expression was exclusively detected in splenic DC cells of homozygous CD40 humanized mice (B-hCD40) and was absent in wild-type controls) (B).
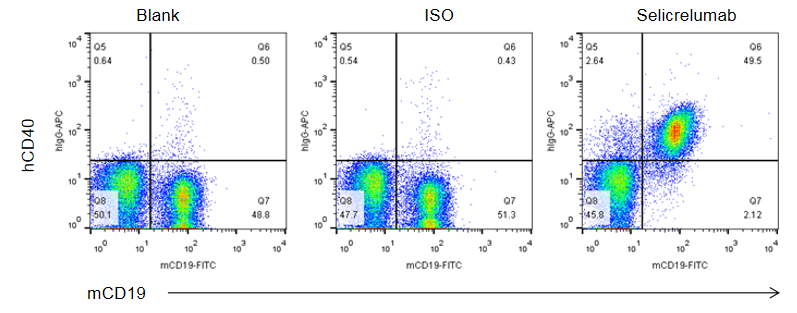
Binding of anti-human CD40 antibodies to B cells was evalsuated in CD40 humanized mice (B-hCD40) by flow cytometry. Splenocytes were isolated from female B-hCD40 mice (n = 3) and stained for mCD19 and human CD40 expression. Single live cells were first gated on the CD45⁺ population and subsequently analyzed for CD19⁺ B cells. Flow cytometry analysis demonstrated that human CD40 was clearly detectable on CD19⁺ B cells in B-hCD40 mice, as evidenced by specific binding of the anti-human CD40 antibody selicrelumab (in-house), whereas minimal signal was observed in the blank and isotype control groups.
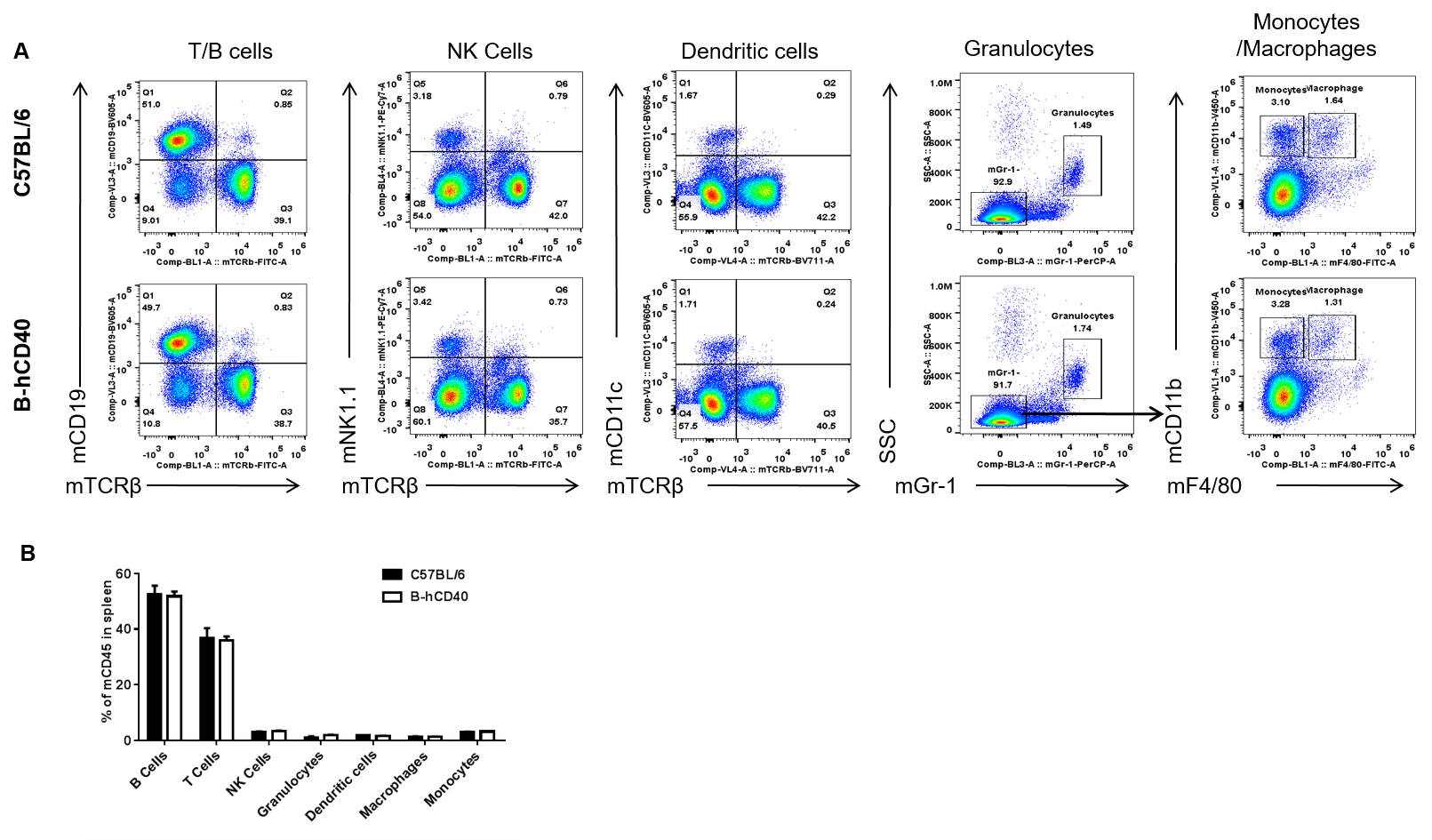
Analysis of spleen leukocyte subpopulations by FACS. Splenocytes were isolated from female C57BL/6 and B-hCD40 mice (n=3). Flow cytometry analysis of the splenocytes was performed to assess leukocyte subpopulations. A. Representative FACS plots. Single live cells were gated for CD45 population and used for further analysis as indicated here. B. Results of FACS analysis. Percent of T, B, NK, Monocyte, DC and macrophage cells in homozygous B-hCD40 mice were similar to those in the C57BL/6 mice, demonstrating that introduction of hCD40 in place of its mouse counterpart does not change the overall development, differentiation or distribution of these cell types in spleen.
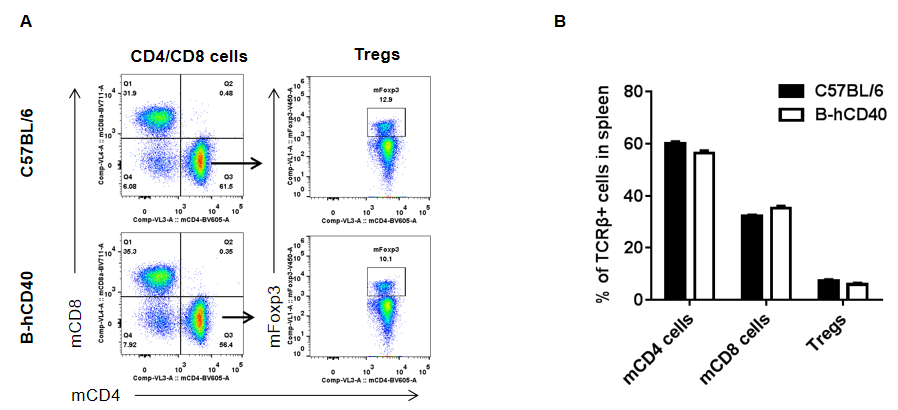
Strain-specific analysis of spleen T cell subpopulations in CD40 humanized mice (B-hCD40) by flow cytometry. Splenocytes were isolated from female wild-type C57BL/6 mice and CD40 humanized mice (B-hCD40) (n = 3) and analyzed by flow cytometry to characterize major T cell subsets in the spleen. (A) Representative FACS plots illustrate the gating strategy used to identify CD3⁺ T cells, followed by discrimination of CD4⁺ T cells, CD8⁺ T cells, and regulatory T cells (Tregs, Foxp3⁺). Single live CD45⁺ cells were gated prior to downstream T cell subset analysis, as indicated. (B) Quantitative analysis of T cell subpopulations shows that the proportions of CD4⁺ T cells, CD8⁺ T cells, and Tregs in the spleen of CD40 humanized mice (B-hCD40) are comparable to those observed in wild-type C57BL/6 mice. These results demonstrate that replacement of mouse CD40 with human CD40 does not alter the overall development, differentiation, or distribution of major T cell subpopulations in the spleen, supporting the immunological integrity of the CD40 humanized mice (B-hCD40) model.
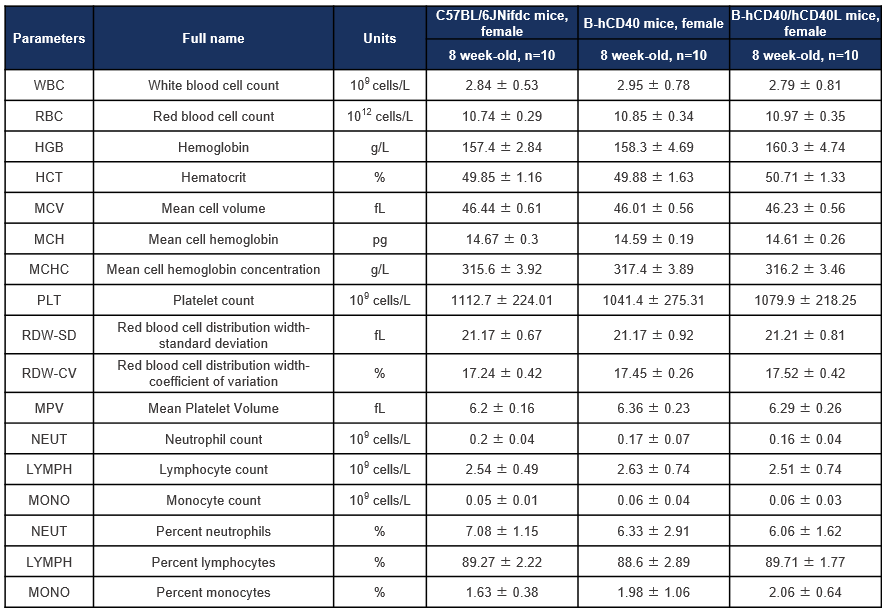
Hematology analysis of CD40 humanized mice (B-hCD40) and CD40/CD40L double-humanized mice (B-hCD40/hCD40L). Complete blood count (CBC) parameters were assessed in female wild-type (C57BL/6JNifdc), CD40 humanized mice (B-hCD40), and CD40/CD40L double-humanized mice (B-hCD40/hCD40L) at 8 weeks of age. Values are expressed as mean ± SD.
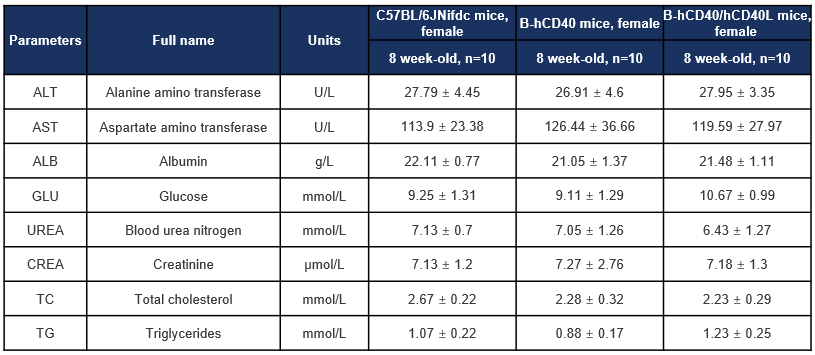
Biochemical analysis was performed to evalsuate key serum biochemical parameters in CD40 humanized mice (B-hCD40) and CD40/CD40L double-humanized mice (B-hCD40/hCD40L). Female wild-type C57BL/6JNifdc mice, B-hCD40 mice, and B-hCD40/hCD40L mice (8 weeks old, n = 10 per group) were included in the study. Serum samples were collected and analyzed for liver function markers (ALT, AST, ALB), metabolic indicators (glucose, total cholesterol, triglycerides), and renal function parameters (blood urea nitrogen and creatinine). Values are expressed as mean ± SD.
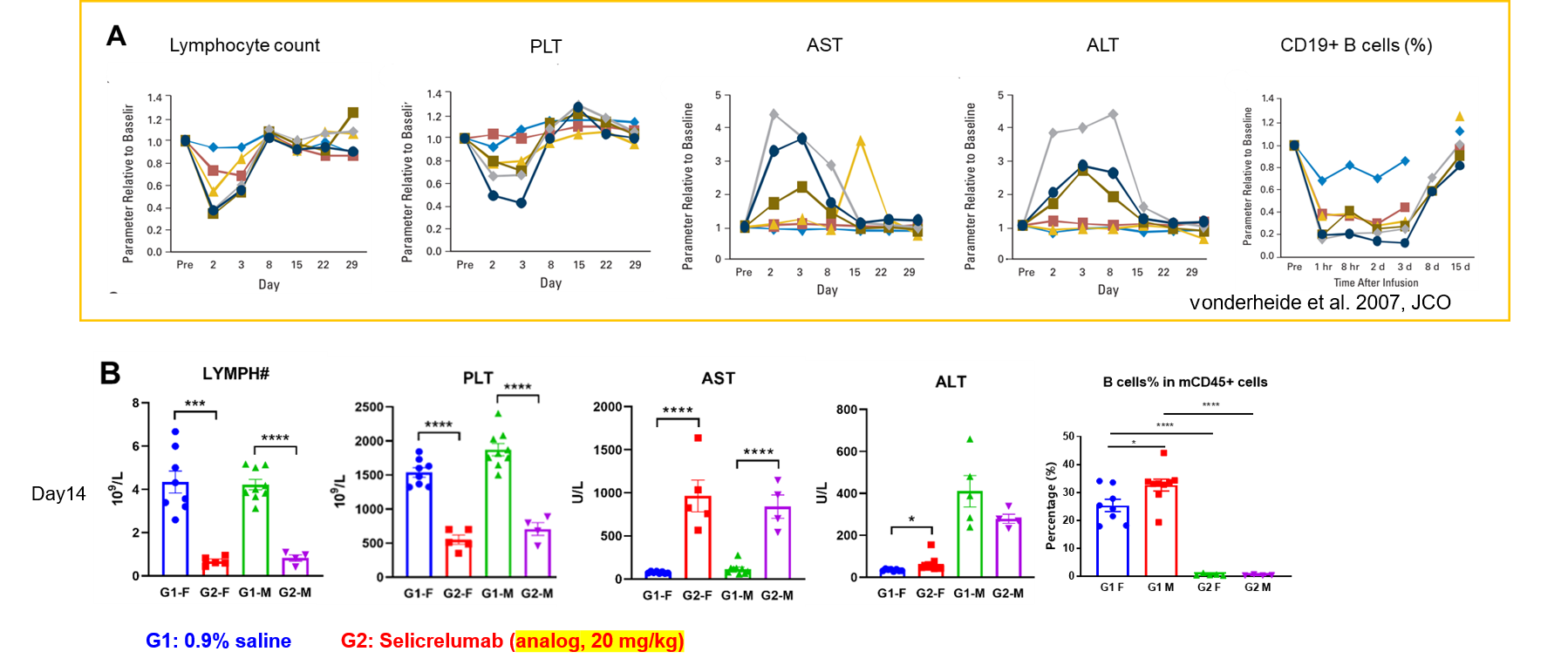
Immune-related adverse event comparison between patients and humanized B-hCD40 mice.(A) After injection of selicrelumab, the clinical patients had obvious hematotoxicity, and the number of peripheral lymphocytes and platelets in the blood decreased. The increase of ALT and AST suggested the occurrence of hepatotoxicity. In addition, CD19+ B cells decreased significantly. (B) B-hCD40 mice of different genders were treated with Selicrelumab, which showed adverse reactions consistent with clinical symptoms.
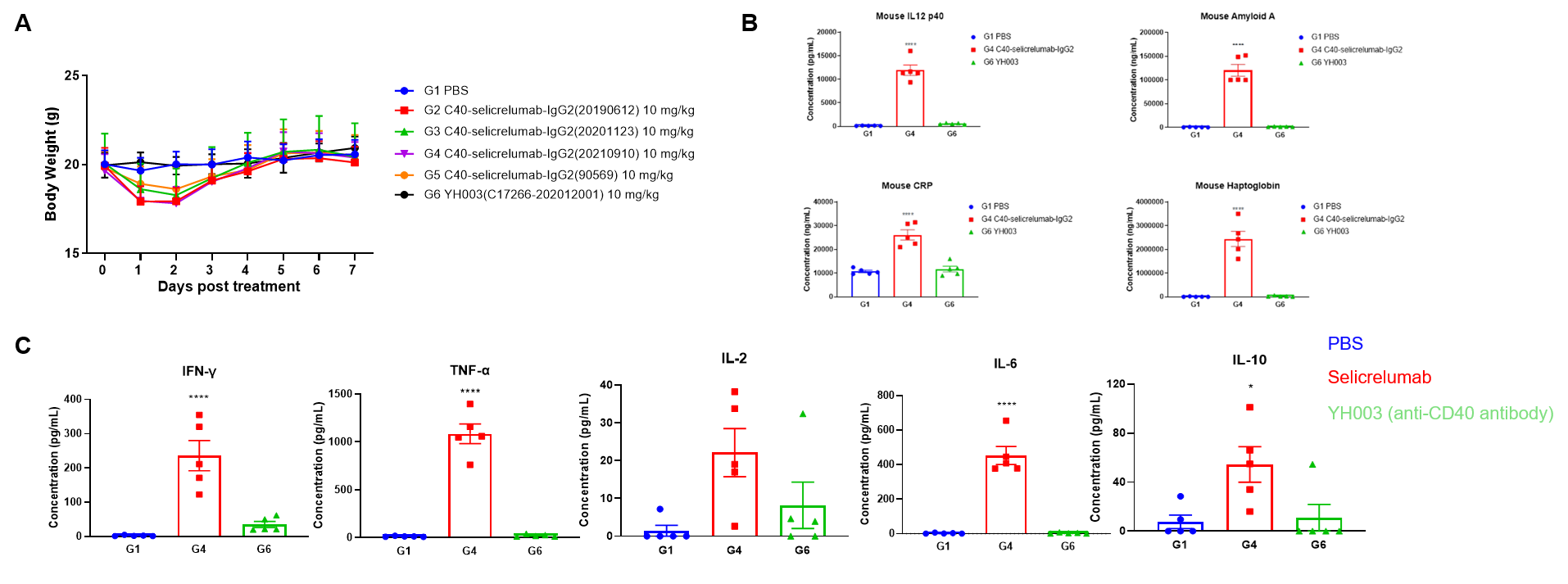
Toxicological profiles of Selicrelumab (analog) in B-hCD40 mice. (A) The body weight of mice in selicrelumab treatment group decreased significantly. (B) Acute phase proteins such as CRP, Amyloid A, Haptoglobin and IL12P40 is increased in response to Selicrelumab in B-hCD40 mice. (C) The mice treated with selicrelumab had obvious cytokine release syndrome, IFN- γ, TNF- α, IL-2, IL-6, IL-10 increased obviously.
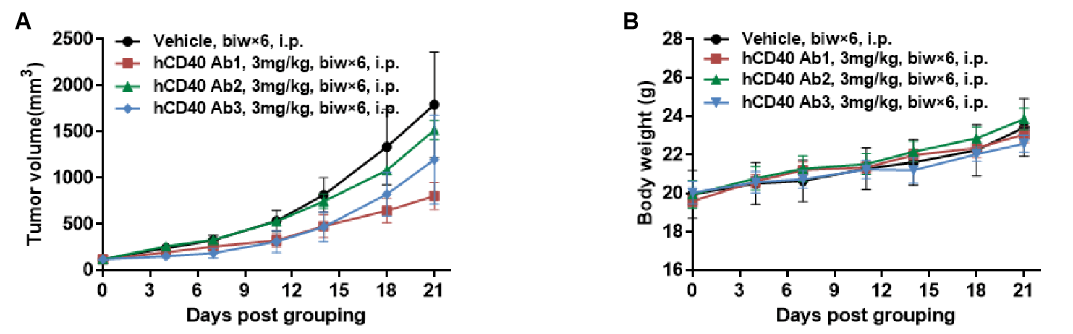
Antitumor activity of anti-human CD40 antibodies in CD40 humanized mice (B-hCD40). Murine colon carcinoma MC38 cells (5 × 10⁵) were subcutaneously implanted into heterozygous CD40 humanized mice (B-hCD40, female, 4 weeks old, n = 5). Mice were randomized when tumor volume reached approximately 100 mm³ and subsequently treated with three anti-human CD40 antibodies (3 mg/kg, biw × 6, i.p.) or vehicle control, as indicated. (A) Tumor growth inhibition. Treatment with anti-human CD40 antibodies significantly suppressed MC38 tumor growth in B-hCD40 mice compared with the vehicle group, demonstrating robust in vivo antitumor efficacy. (B) Body weight monitoring. No significant body weight loss was observed across treatment groups during the study period, indicating acceptable tolerability of anti-human CD40 antibody treatment. Collectively, these results demonstrate that CD40 humanized mice (B-hCD40) provide a robust and translationally relevant in vivo preclinical model for evalsuating the antitumor efficacy and safety of anti-human CD40 therapeutic antibodies. Values are expressed as mean ± SEM.
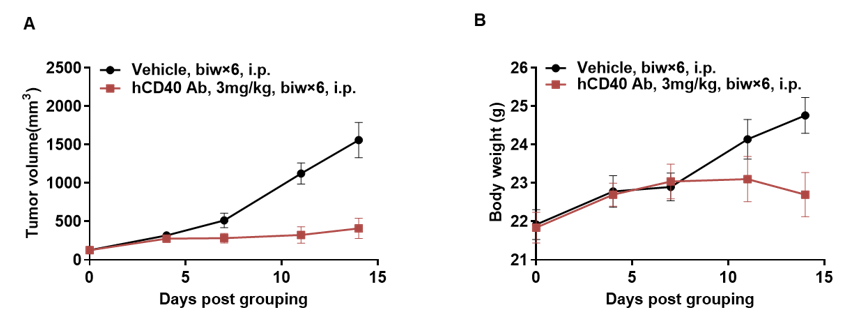
Antitumor activity of anti-human CD40 antibodies in CD40 humanized mice (B-hCD40). Murine colon carcinoma MC38 cells (5 × 10⁵) were subcutaneously implanted into heterozygous CD40 humanized mice (B-hCD40, female, 4 weeks old, n = 5). Mice were randomized when tumor volume reached approximately 100 mm³ and subsequently treated with three anti-human CD40 antibodies (3 mg/kg, biw × 6, i.p.) or vehicle control, as indicated. (A) Tumor growth inhibition. Treatment with anti-human CD40 antibodies significantly suppressed MC38 tumor growth in B-hCD40 mice compared with the vehicle group, demonstrating robust in vivo antitumor efficacy. (B) Body weight monitoring. No significant body weight loss was observed across treatment groups during the study period, indicating acceptable tolerability of anti-human CD40 antibody treatment. Collectively, these results demonstrate that CD40 humanized mice (B-hCD40) provide a robust and translationally relevant in vivo preclinical model for evalsuating the antitumor efficacy and safety of anti-human CD40 therapeutic antibodies. Values are expressed as mean ± SEM.
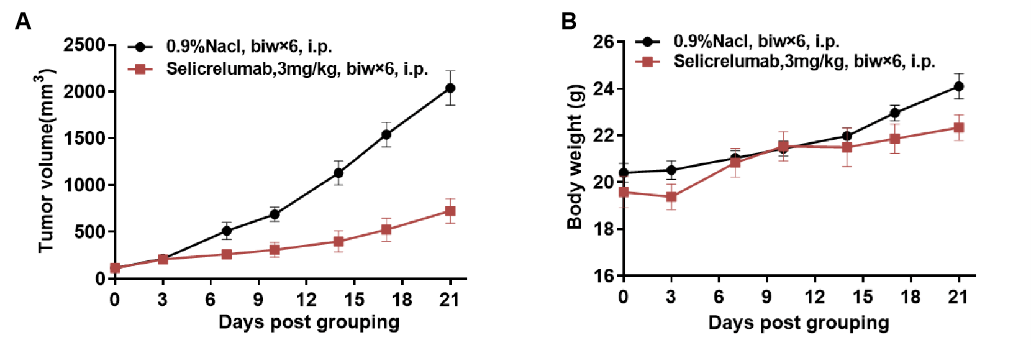
Antitumor activity of anti-human CD40 antibodies in CD40 humanized mice (B-hCD40). Murine colon carcinoma MC38 cells (5 × 10⁵) were subcutaneously implanted into homozygous CD40 humanized mice (B-hCD40; female, 8–9 weeks old, n = 5). When tumor volumes reached approximately 150 ± 50 mm³, mice were randomized and treated with the anti-human CD40 antibody Selicrelumab (in-house, 3 mg/kg, biweekly × 6, i.p.) or vehicle control (0.9% NaCl). (A) Tumor growth inhibition. Treatment with Selicrelumab resulted in a marked suppression of MC38 tumor growth compared with the vehicle group, demonstrating effective antitumor activity mediated through human CD40 signaling in B-hCD40 mice. (B) Body weight monitoring. These results demonstrate that CD40 humanized mice (B-hCD40) provide a robust and translationally relevant preclinical model for in vivo evalsuation of anti-human CD40 therapeutic antibodies. Values are expressed as mean ± SEM.
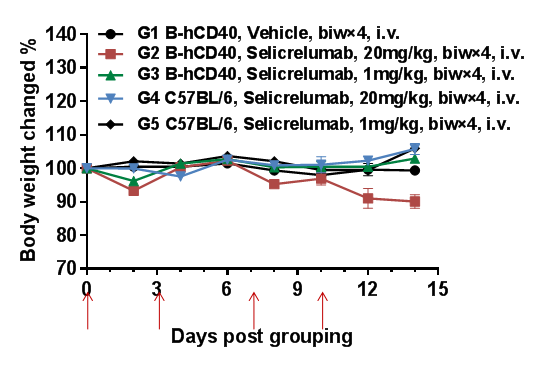
High-dose selicrelumab (anti-human CD40) induces moderate body weight reduction in CD40 humanized mice (B-hCD40), but not in wild-type C57BL/6 mice.
To evalsuate the tolerability of anti-human CD40 antibody treatment, homozygous CD40 humanized mice (B-hCD40) and wild-type C57BL/6 mice were treated with vehicle or selicrelumab (in-house) according to the dosing schedule indicated by red arrows (n = 3). Selicrelumab was administered intravenously at doses of 1 mg/kg or 20 mg/kg, twice weekly for four weeks. In CD40 humanized mice (B-hCD40), treatment with selicrelumab at 20 mg/kg resulted in a moderate reduction in body weight compared with vehicle control, whereas the same dosing regimen had no observable effect on body weight in wild-type C57BL/6 mice. Lower-dose selicrelumab (1 mg/kg) did not cause significant body weight changes in either genotype. Values are expressed as mean ± SEM.
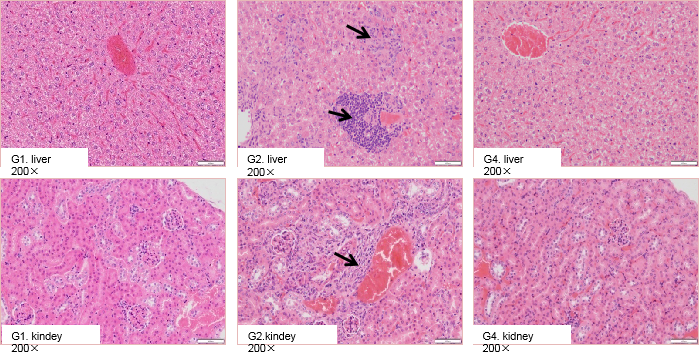
High-dose selicrelumab treatment induced pathological changes in the liver and kidney of CD40 humanized mice (B-hCD40). Homozygous B-hCD40 mice and wild-type C57BL/6 mice were treated with PBS or selicrelumab (n = 3), and tissue pathology analysis was performed on day 14 after treatment. In CD40 humanized mice (B-hCD40) mice, selicrelumab administered at 20 mg/kg resulted in increased lymphocyte infiltration in both liver and kidney tissues, as indicated by arrows. In contrast, the same treatment did not induce detectable pathological changes in the liver or kidney of wild-type C57BL/6 mice.
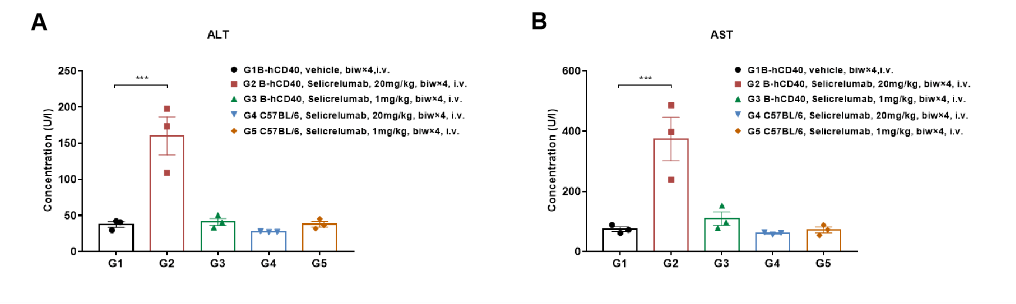
High-dose selicrelumab treatment resulted in significant changes in serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels in CD40 humanized mice (B-hCD40). Homozygous B-hCD40 mice and wild-type C57BL/6 mice were treated with PBS or selicrelumab (in-house) according to the indicated dosing schedule (n = 3). Blood biochemical analysis was performed 24 hours after termination of the efficacy evalsuation experiment. (A) Serum alanine aminotransferase (ALT) levels.(B) Serum aspartate aminotransferase (AST) levels.
In B-hCD40 mice, selicrelumab administered at 20 mg/kg caused a significant increase in ALT and AST levels (A, B) compared with PBS-treated controls, but not in wild-type C57BL/6 mice. These findings indicate a target-dependent liver toxicity profile associated with high-dose anti-human CD40 antibody treatment. Values are expressed as mean ± SEM.
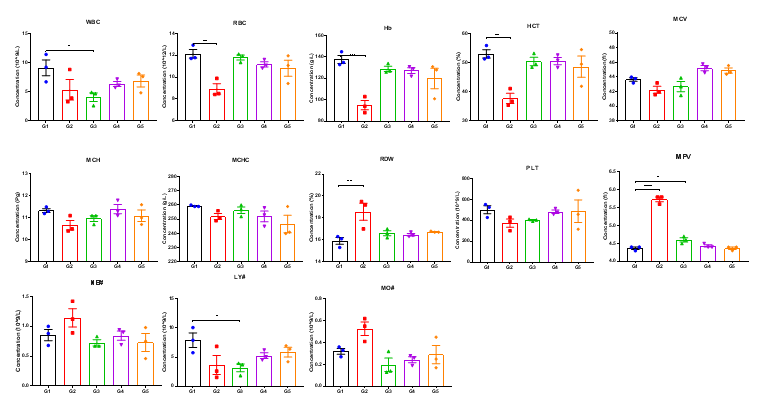
High-dose human CD40 antibody treatment induced significant alterations in hematological parameters in CD40 humanized mice (B-hCD40). Homozygous CD40 humanized mice (B-hCD40) and wild-type C57BL/6 mice were treated with PBS or selicrelumab (in-house) as indicated in the graph (n = 3). Complete blood count (CBC) analysis was performed 24 hours after the last dose on day 11. In CD40 humanized mice (B-hCD40) , selicrelumab administered at 20 mg/kg resulted in a significant reduction in red blood cell count (RBC) and hemoglobin (Hb) levels, accompanied by decreases in hematocrit (HCT) and lymphocyte percentage (LY#), as well as increases in red cell distribution width (RDW) and mean platelet volume (MPV), compared with PBS-treated controls. In contrast, the same treatment regimen did not induce significant changes in CBC parameters in wild-type C57BL/6 mice. Values are expressed as mean ± SEM.
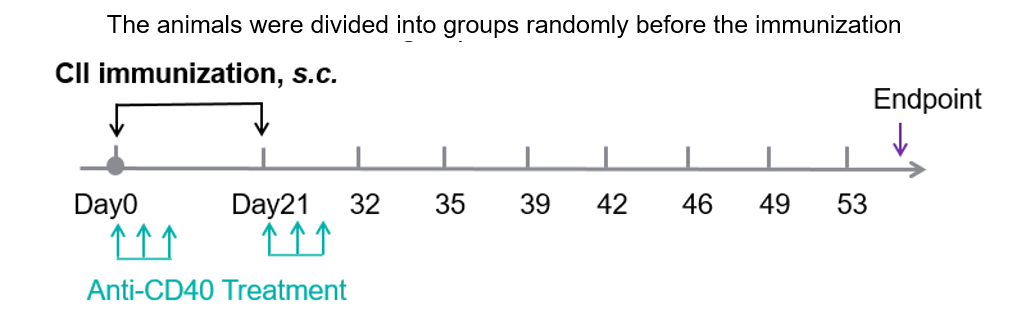
Experimental schedule for induction of CIA and in vivo efficacy of anti-human CD40 antibody.
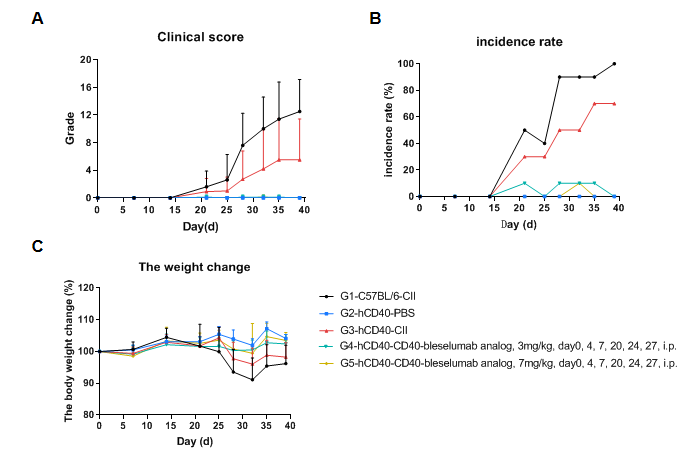
In vivo Efficacy of anti-Human CD40 antibody in a collagen-induced arthritis (CIA) model using CD40 humanized mice (B-hCD40). Mice were randomly assigned to treatment groups prior to immunization. CIA was induced by subcutaneous immunization with type II collagen (CII). Anti-human CD40 antibody treatment was administered according to the indicated schedule, and animals were monitored until the experimental endpoint. (A) Clinical score: Mice in the CIA model groups (G1 & G3) exhibited a progressive increase in clinical arthritis scores, confirming successful induction of the CIA model. Treatment with the anti-human CD40 antibody (G4 & G5) significantly reduced clinical scores compared with the untreated model group, indicating effective disease control. (B) Disease incidence: The incidence of arthritis increased markedly in the model group, whereas anti-human CD40 antibody treatment resulted in a significantly lower maximum incidence rate, demonstrating a protective therapeutic effect. (C) Body weight change: Body weight fluctuations were more pronounced in the CIA model group than in non-model controls. Anti-human CD40 antibody–treated mice showed improved weight stability during the study period. These results demonstrate that anti-human CD40 antibody treatment effectively alleviates disease severity and reduces incidence in the CIA model established in CD40 humanized mice (B-hCD40), supporting the utility of this model for in vivo preclinical evalsuation of anti-human CD40 therapeutics.
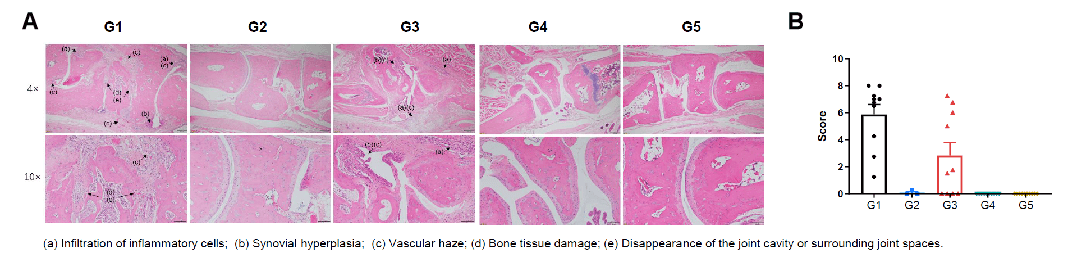
Pathological evalsuation of anti-human CD40 antibody bleselumab in a CIA model using CD40 humanized mice (B-hCD40). Histopathological analysis was conducted to evalsuate the therapeutic efficacy of the anti-human CD40 antibody bleselumab (in-house) in a collagen-induced arthritis (CIA) model established in CD40 humanized mice (B-hCD40). (A) Representative ankle joint sections from B-hCD40 mice were stained with H&E. (B) Pathological scores were quantitatively assessed. In the non-model control group (G2), CD40 humanized mice (B-hCD40) showed no significant pathological abnormalities, with smooth cartilage surfaces and clearly defined joint cavities. In contrast, mice in the CIA model group (G1 & G3) exhibited typical arthritis-associated pathological features, including infiltration of inflammatory cells, synovial hyperplasia, and vascular haze. Compared with the model group, bleselumab-treated groups (G4 & G5) in CD40 humanized mice (B-hCD40) showed marked attenuation of inflammatory cell infiltration and synovial hyperplasia, accompanied by significant improvement in pathological scores. These findings demonstrate that bleselumab effectively alleviates arthritis-related pathological damage in CD40 humanized mice (B-hCD40), supporting the translational relevance of this model for in vivo evalsuation of anti-human CD40 therapies in inflammatory arthritis.
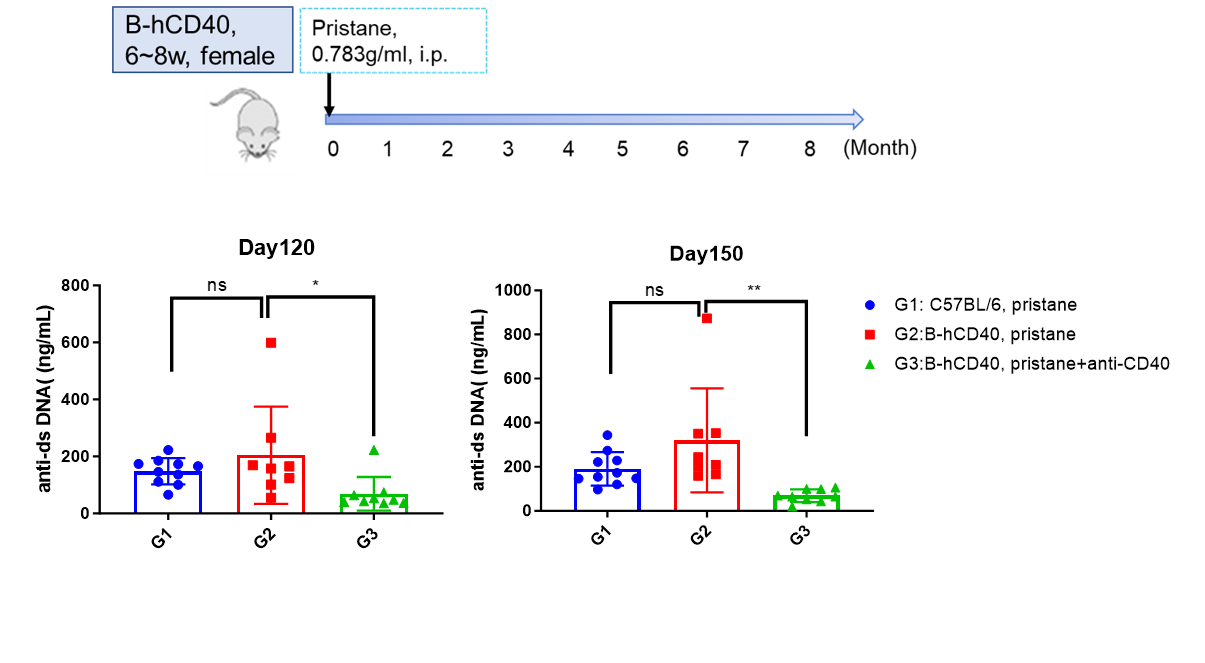
Establishment of SLE Mouse Model based on B-hCD40 Mice. Content of mouse Anti-dsDNA IgG in serum was decreased after treatment with anti-CD40 antibody in pristane-induced SLE B-hCD40 Mouse Model.